Allosteric-like FAST biosensors for glutamate, K+, ATP, just disclosed in ACS Sensors 2023. A work led by Prof. Arnaud Gautier of Sorbonne University, and a co-founder of The Twinkle Factory.
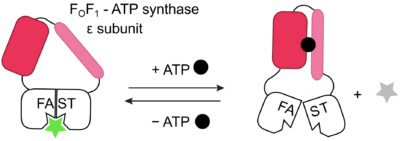
Inspired by allosteric proteins, they designed fluorescent sensors responding to biological analytes through the incorporation of various sensing units into the protein tag FAST. FAST is indeed known to bind cognate fluorogens, dark in their free form, fluorescent once bound. As a result,onformational coupling between FAST and a sensing domain can generate fluorescent sensors with allosteric-like behavior in which fluorogen binding and fluorescence, is conditioned to analyte recognition.
Yet, one can engineer FAST into two complementary fragments, splitFAST, or circularly permuted versions, cpFAST. As a result FAST opens a variety of topological designs for optimizing the conformational coupling with the sensing units. Moreover, one can modulate the dynamic range through (i) a change in the fluorogen concentration, (ii) the use of fluorogens with different binding affinities, or (iii) the use of FAST variants with different fluorogen affinities.
Finally, the authors have designed sensors for analytes as different as glutamate, K+, and ATP.
“Future efforts should focus on (i) the implementation of such sensors for the optical monitoring of analytes of interest in live cells and organisms in real time and (ii) the development of fluorescent or bioluminescent assays for the monitoring of metabolites at the point of care for the diagnostic and monitoring of diseases.”
Fanny Broch et al.
The Twinkle Factory offers commercial fluorogens for FAST, pFAST, greenFAST & redFAST, frFAST, and splitFAST, their related split version. tfFox-NP, a bright non-permeant fluorogen, and tfDarth, a quencher, have recently completed the product range.
Read more about FAST biosensors
Engineering of Tunable Allosteric-like Fluorogenic Protein Sensors. Broch, F., El Hajji, L., Pietrancosta, N., & Gautier, A. ACS Sensors 2023.
Circularly permuted fluorogenic proteins for the design of modular biosensors. Tebo, A. G., Pimenta, F. M., Zoumpoulaki, M., Kikuti, C., Sirkia, H., Plamont, M. A., … & Gautier, A. ACS Chemical Biology 2018.
Recent Comments