Looking after bacterial protein export? A team led by Prof. Christophe Beloin of Institut Pasteur and Prof. Nicolas Desprat of ENS has just disclosed FAST for monitoring protein trafficking at Listeria surface in Scientific Reports 2020.
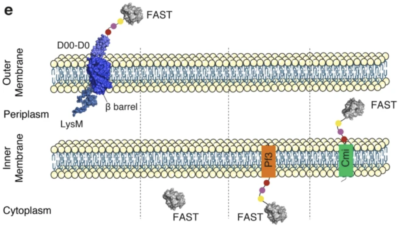
Bacterial proteins exported to the cell surface play key cellular functions. For example, surface proteins such as adhesins, transporters or hydrolases are of the utmost interest. However, monitoring their dynamics in live imaging remains challenging. This is indeed due to the limited availability of fluorescent probes remaining functional after secretion. In this work, the authors used the Escherichia coli intimin and the Listeria monocytogenes InlB invasin as surface-exposed scaffolds fused with FAST. FAST is indeed the breakthrough chemogenetic fluorescent reporter promoted by The Twinkle Factory. They further used a combination of membrane-permeant (HBR-3,5DM) and non‐permeant (HBRAA-3E) fluorogens to reveal FAST tags. Both indeed fluoresce only once binding FAST. The authors hence demonstrated that FAST exposed at the bacterial surface can be fully functional. Moreover, one can specifically tag the external side of the bacterial envelop in both diderm and monoderm bacteria.
As a result, this work opens new avenues to study the organization and dynamics of the bacterial cell surface proteins. Moreover, it adds Listeria to the array of bacteria successfully engineered with FAST: Clostridium, Escherichia.
The Twinkle Factory distributes related commercial fluorogens under the tradenames tfAmber (HBR-3,5DM) and tfAmber-NP (HBRAA-3E).
Stain different, tag FAST!
More readings on FAST and bacterial proteins
-
“Visualizing the dynamics of exported bacterial proteins with the chemogenetic fluorescent reporter FAST”, Sci. Rep. 2020
-
“A Strongly Fluorescing Anaerobic Reporter and Protein-Tagging System for Clostridium Organisms Based on the Fluorescence-Activating and Absorption-Shifting Tag Protein (FAST)”, Appl. Environ. Microbiol. 2019
- “The inducible chemical-genetic fluorescent marker FAST outperforms classical fluorescent proteins in the quantitative reporting of bacterial biofilm dynamics”, Sci. Rep. 2018
Recent Comments